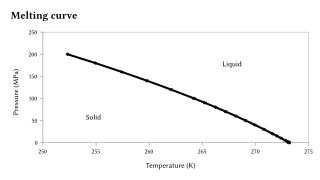
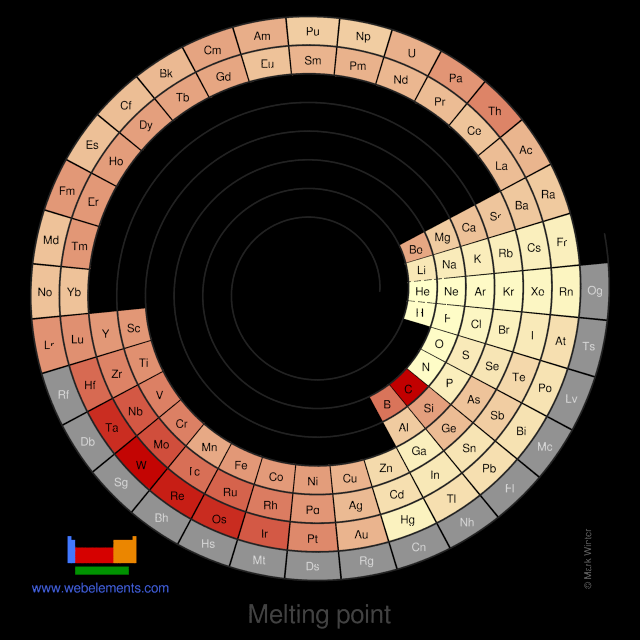
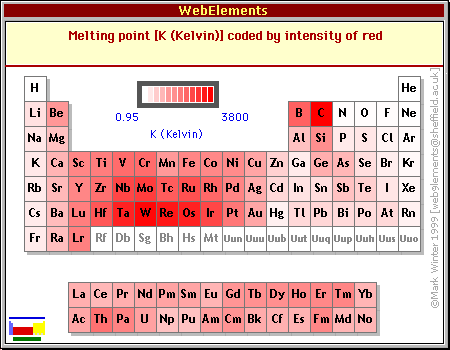
PDF) Approximated equations for molar volumes of pure solid fcc metals and their liquids from zero Kelvin to above their melting points at standard pressure

Vecteur Stock Temperature scales showing differences between Kelvin, Celsius or centigrade, and Fahrenheit scale. Boiling point of water, freezing point of water, absolute zero. Set of three thermometers isolated. | Adobe Stock

PDF) Approximated equations for molar volumes of pure solid fcc metals and their liquids from zero Kelvin to above their melting points at standard pressure

Peak temperatures experienced by iron micrometeoroids in the Martian... | Download Scientific Diagram
How is aluminium melted at a lower temperature than its melting point (i.e. 660.3 degree Celsius) when mixed in small proportions to zinc? - Quora