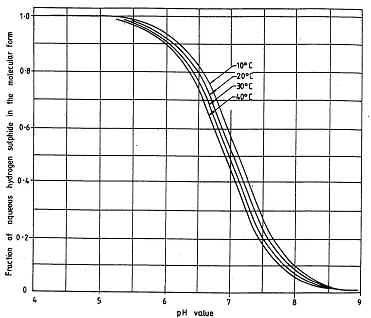
Low-Temperature Oxidation of Hydrogen Sulfide and Formaldehyde Pollutants in Humid Air by UV Radiation at 184.95 and 253.65 nm | The Journal of Physical Chemistry A

Energies | Free Full-Text | Methane and Hydrogen Sulfide Production from Co-Digestion of Gummy Waste with a Food Waste, Grease Waste, and Dairy Manure Mixture

High pressure adsorption of hydrogen sulfide and regeneration ability of ultra-stable Y zeolite for natural gas sweetening - ScienceDirect
New catalyst can turn smelly hydrogen sulfide into a cash cow | Rice News | News and Media Relations | Rice University

Inhibition and Promotion of Pyrolysis by Hydrogen Sulfide (H2S) and Sulfanyl Radical (SH) | The Journal of Physical Chemistry A

Hydrogen sulfide mole fraction. Flame condition: H 2 /O 2-N 2 with 100%... | Download Scientific Diagram
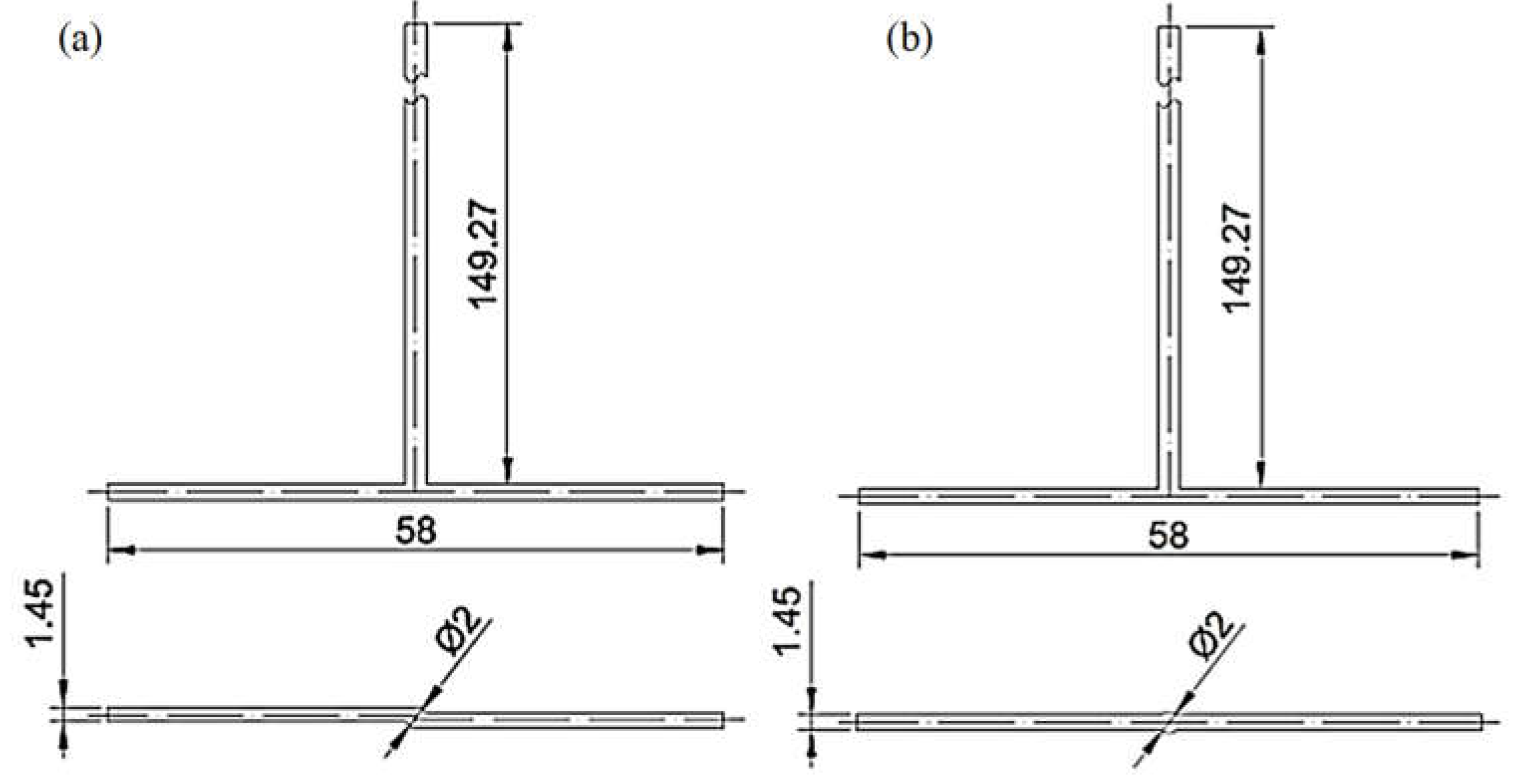
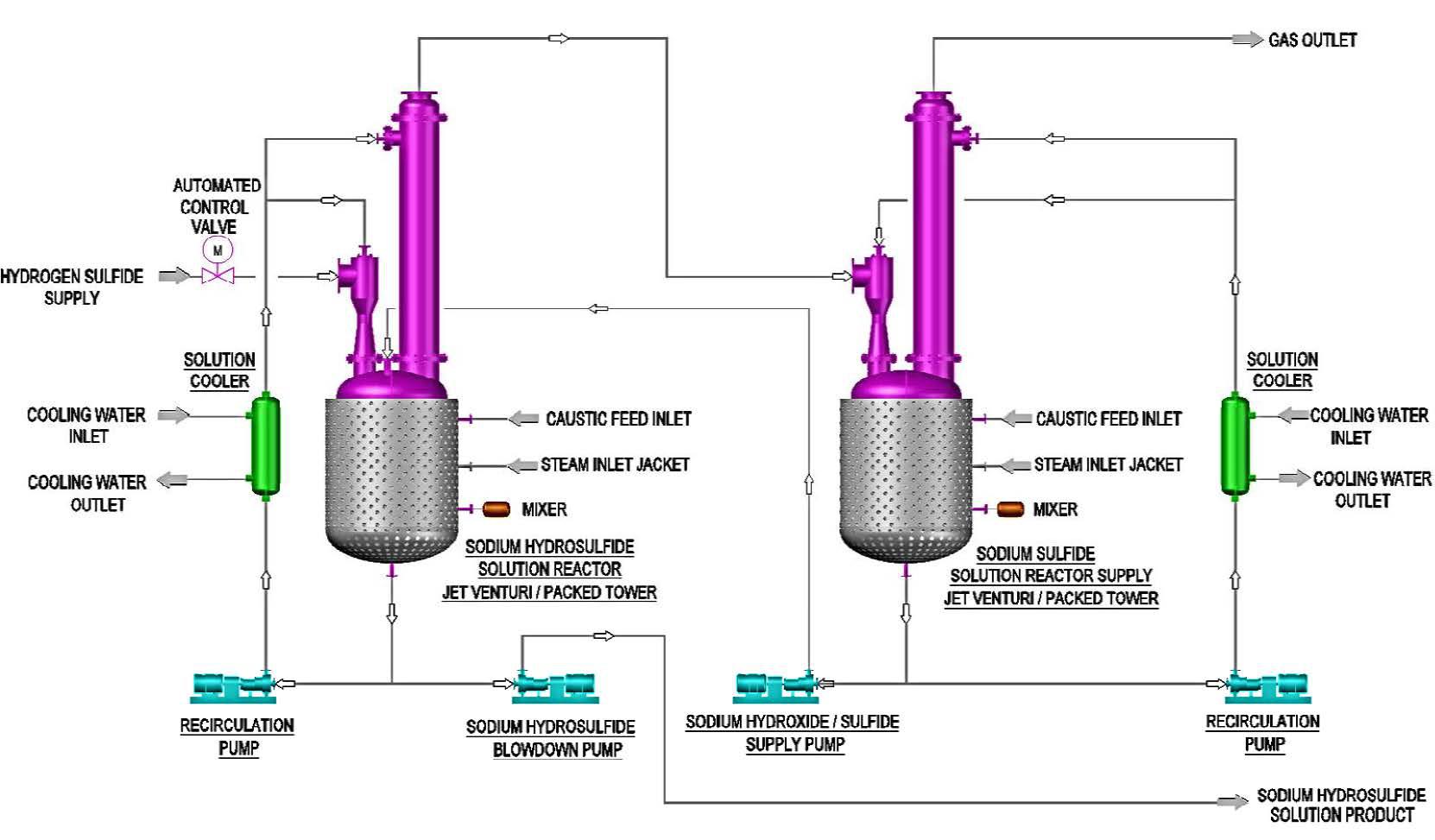
Applied Sciences | Free Full-Text | Influence of Process Parameters and Reducing Agent on the Size of MoS2 Nanoparticles Obtained in Impinging Jet Reactor

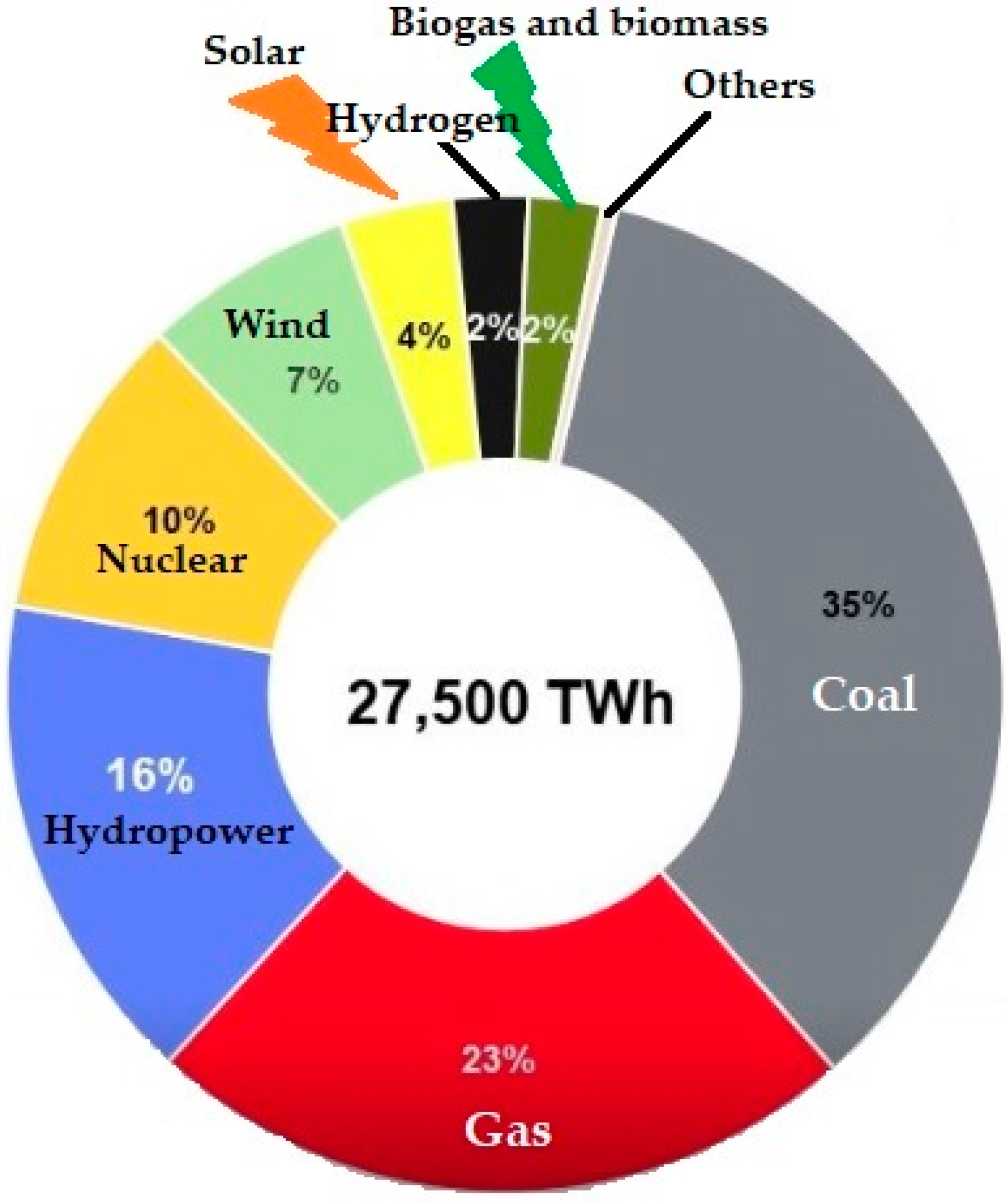
Energies | Free Full-Text | Warm Plasma Application in Tar Conversion and Syngas Valorization: The Fate of Hydrogen Sulfide

Reaction of Hydrogen Sulfide with a Chlorine Atom in the Temperature Range of 273 to 366 K | SpringerLink
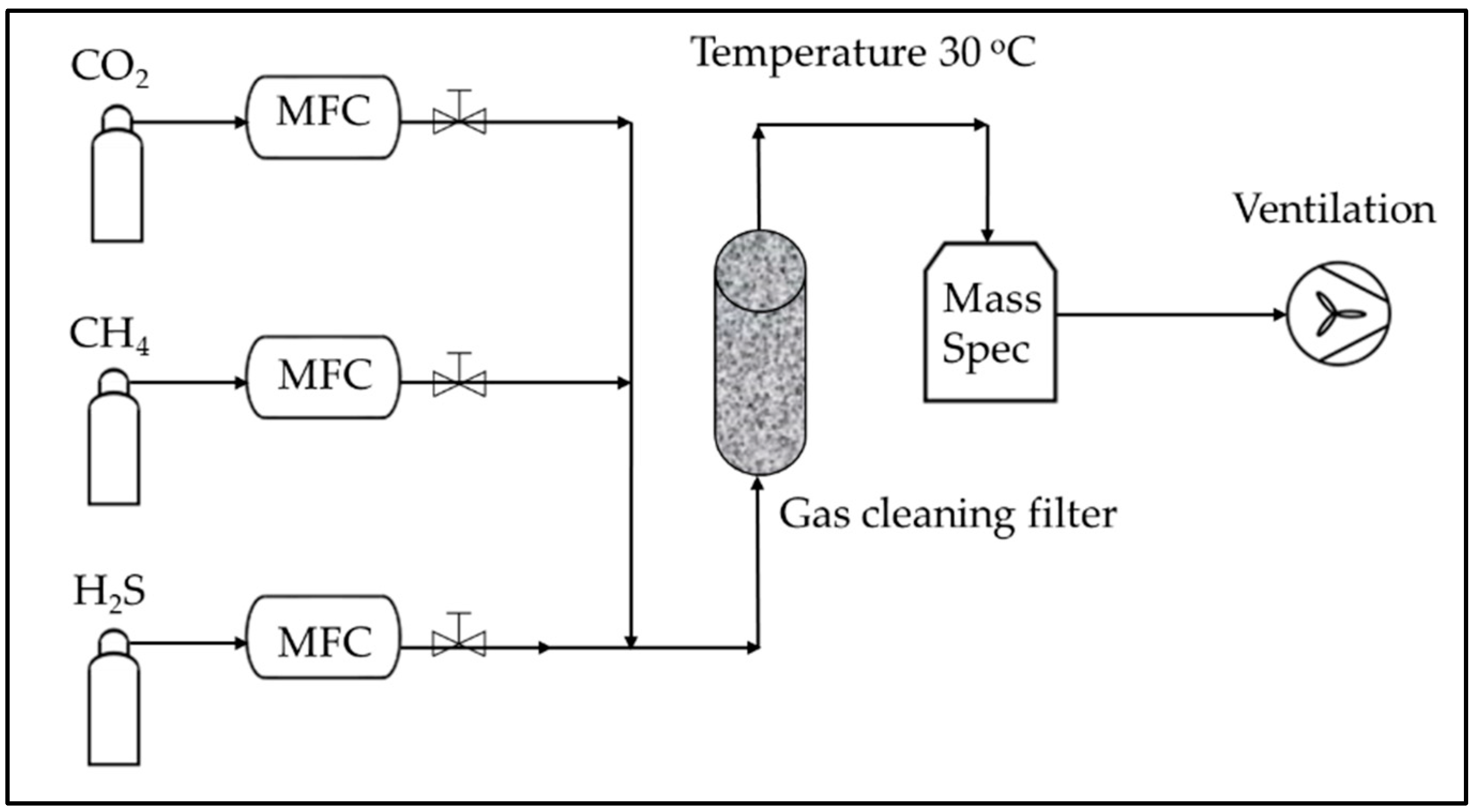
Processes | Free Full-Text | Study of H2S Removal Capability from Simulated Biogas by Using Waste-Derived Adsorbent Materials

Effects of Morphology and Surface Properties of Copper Oxide on the Removal of Hydrogen Sulfide from Gaseous Streams | Industrial & Engineering Chemistry Research