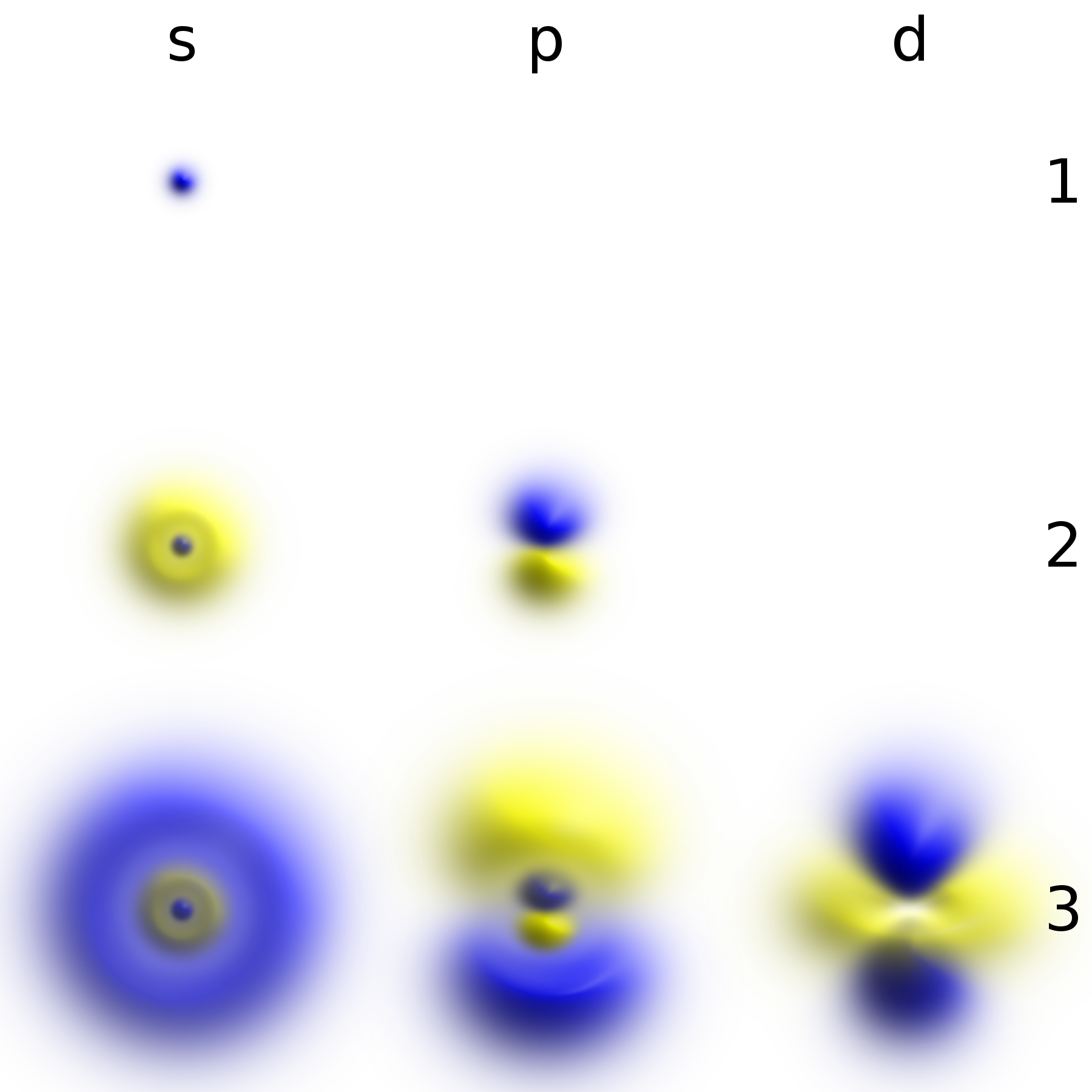
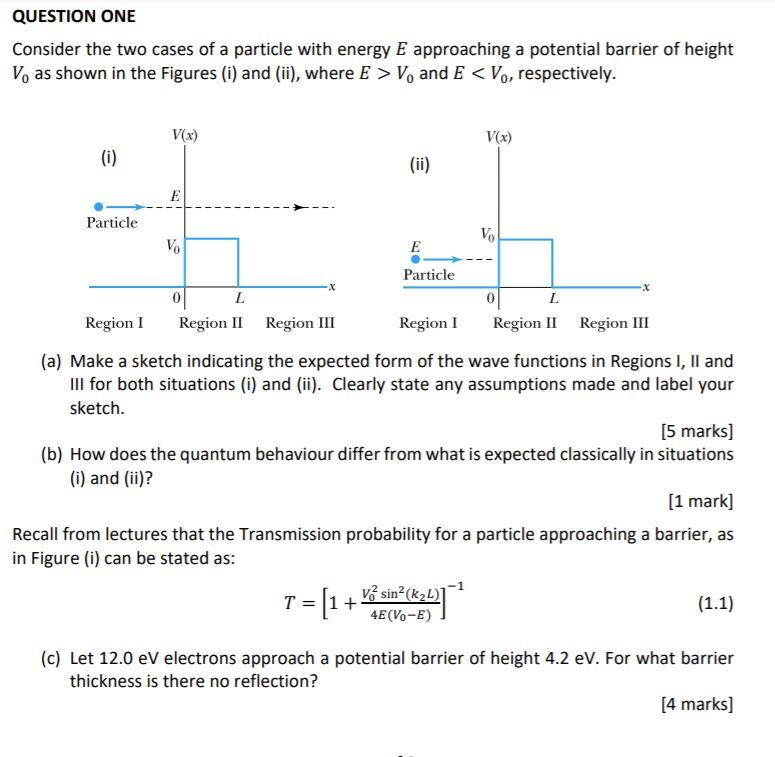
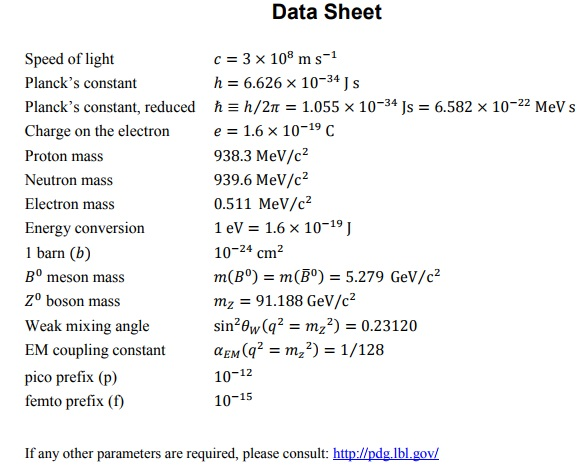
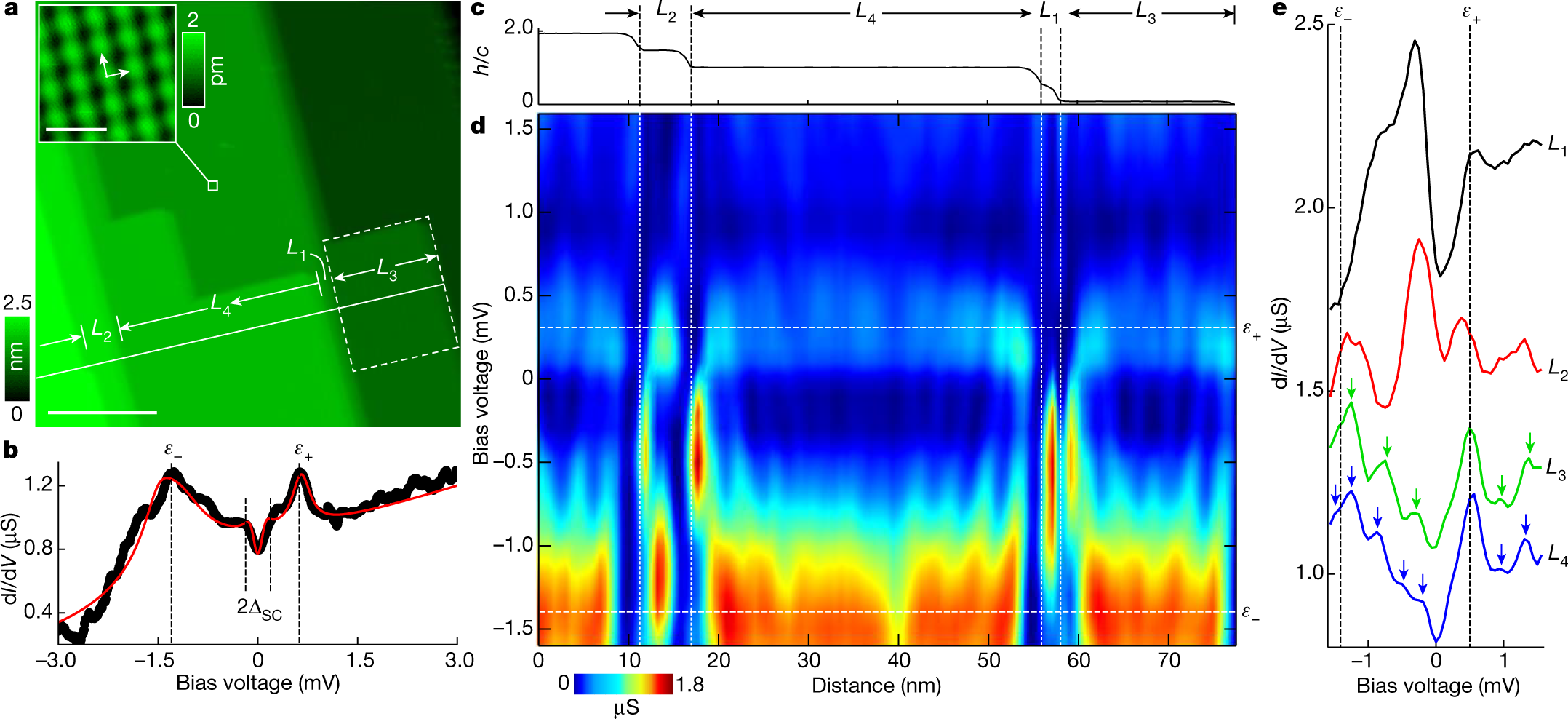
Toward Detection of the Molecular Parity Violation in Chiral Ru(acac)3 and Os(acac)3 | The Journal of Physical Chemistry Letters

Given: The mass of electron is `9.11 � 10^(�31)`Kg Planck constant is `6.626 �10^(�34)` is:- - YouTube
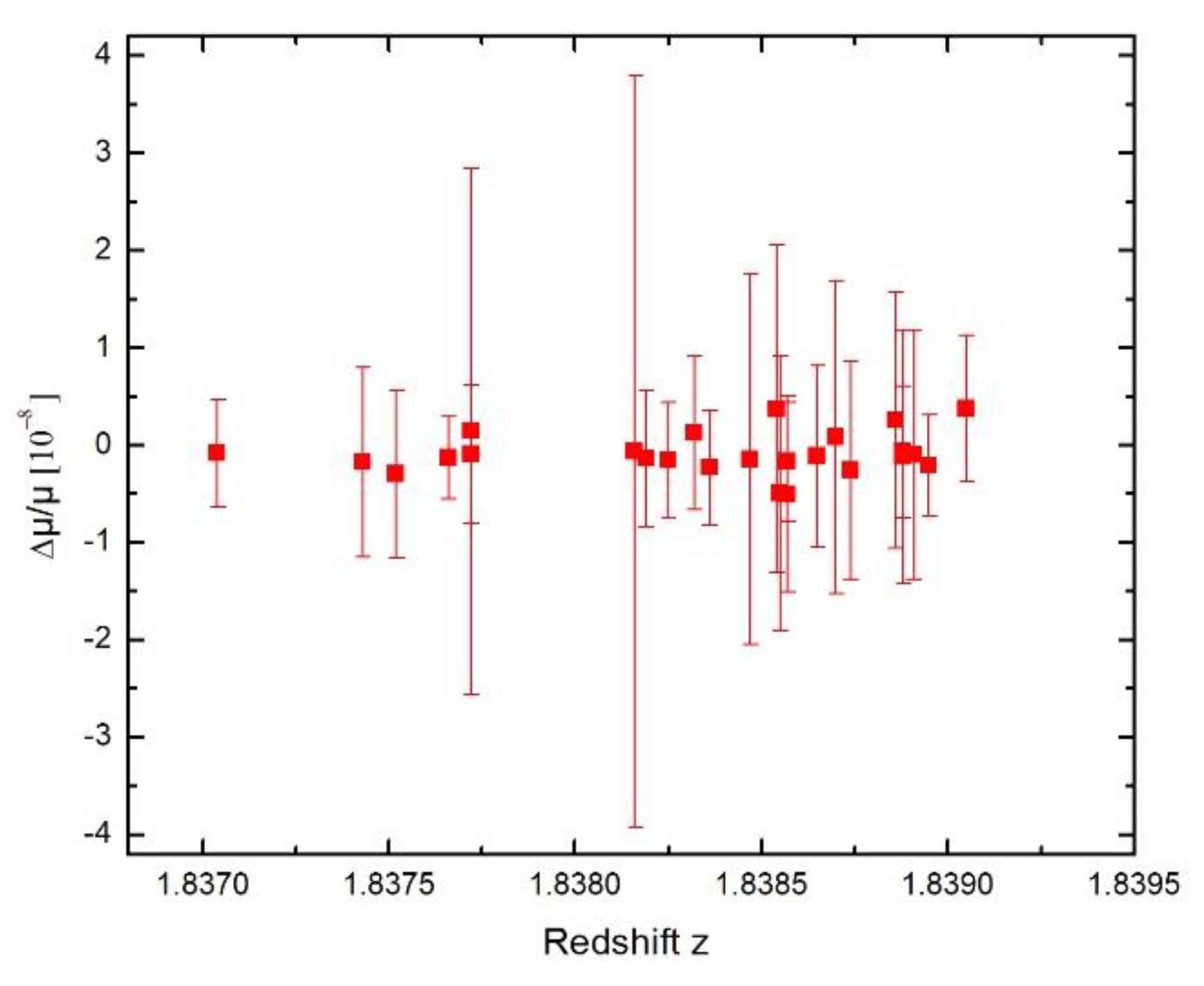
Symmetry | Free Full-Text | New Limit on Space-Time Variations in the Proton-to-Electron Mass Ratio from Analysis of Quasar J110325-264515 Spectra

The mass of electron is 9.11 × 10^-31 kg.Planck's constant is 6.626 × 10^-34 Js then the uncertainty involved in the measurement of velocity within a distance of 0.1 A is :

If `m,e,epsilon_(0) h` and `c` denote mass ,electron , change of electron, plank \'s constant and - YouTube

Synthesis, Electronic Structure, and Electron Transfer Dynamics of (Aryl)ethynyl-Bridged Donor−Acceptor Systems | Journal of the American Chemical Society
AppliedMath | Free Full-Text | Approximate Nonlocal Symmetries for a Perturbed Schrödinger Equation with a Weak Infinite Power-Law Memory
![Calculate the energy of the light having wavelength 45 nm : [Planck's constant h = 6.63 × 10^-34 Js ; speed of light c = 3 × 10^8 ms^-1 ] Calculate the energy of the light having wavelength 45 nm : [Planck's constant h = 6.63 × 10^-34 Js ; speed of light c = 3 × 10^8 ms^-1 ]](https://dwes9vv9u0550.cloudfront.net/images/8714373/032957da-34c2-47ec-a991-027613566c64.jpg)
Calculate the energy of the light having wavelength 45 nm : [Planck's constant h = 6.63 × 10^-34 Js ; speed of light c = 3 × 10^8 ms^-1 ]

SOLVED: Planck's constant is h = 6.626X 10-34J . vacuum is c =3 X 108@ 4.136 x 10-I4eV . S; Speed of light in Elementary charge: € = 1.60 X1O-19 C; mass

Uncertainty in the position of an electron (mass = 9.1 × 10^-31kg) moving with a velocity 300ms^-1 accurate upon 0.001

Physics of electron emission and injection in two‐dimensional materials: Theory and simulation - Ang - 2021 - InfoMat - Wiley Online Library